 The top image shows the CO 2 being trapped beneath a dome, preventing it from migrating laterally or vertically. Diagram depicting two examples of structural trapping. Therefore, the CO 2 will migrate upwards through the porous rocks until it reaches (and is trapped by) an impermeable layer of seal rock. The rock layers and faults within and above the storage formation where the CO 2 is injected act as seals, preventing CO 2 from moving out of the storage formation. Once injected, the supercritical CO 2 can be more buoyant than other liquids present in the surrounding pore space. 
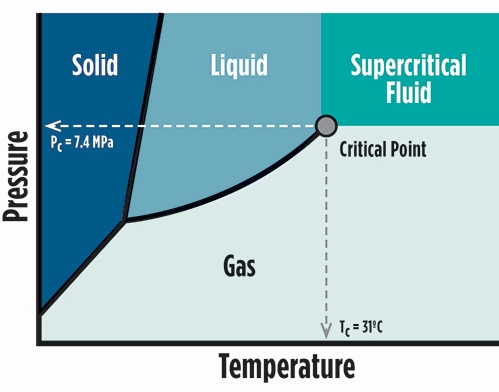
Structural Trapping – Structural trapping is the physical trapping of CO 2 in the rock and is the mechanism that traps the greatest amount of CO 2. The following provides a description of each type of trapping mechanism. Each of these mechanisms plays a role in how the CO 2 remains trapped in the subsurface. There are four main mechanisms that trap the injected CO 2 in the subsurface. Trapping refers to the way in which the carbon dioxide (CO 2) remains underground in the location where it is injected. Reality: The elevated temperatures and pressures that exist at the depths where CO 2 is injected changes its characteristics, allowing for storage of much greater volumes of CO 2 than at the surface. Myth: The CO 2 gas behaves the same in the atmosphere as it does when injected deep underground. This means that CO 2 injected at this depth or deeper will remain in the supercritical condition given the temperatures and pressures present. At depths below about 800 meters (about 2,600 feet), the natural temperature and fluid pressures are in excess of the critical point of CO 2 for most places on Earth. Temperature naturally increases with depth in the Earth’s crust, as does the pressure of the fluids (brine, oil, or gas) in the formations. The main advantage of storing CO 2 in the supercritical condition is that the required storage volume is substantially less than if the CO 2 were at “standard” (room)-pressure conditions. In particular, it is dense like a liquid but has viscosity like a gas. At such high temperatures and pressures, the CO 2 has some properties like a gas and some properties like a liquid. Supercritical CO 2 means that the CO 2 is at a temperature in excess of 31.1☌ (88✯) and a pressure in excess of 72.9 atm (about 1,057 psi) this temperature and pressure defines the critical point for CO 2. The blue numbers show the volume of CO2 at each depth compared to a volume of 100 at the surface.Ĭarbon dioxide (CO 2) can be stored underground as a supercritical fluid. HOW CAN CO 2 BE STORED UNDERGROUND? Illustration of Pressure Effects on CO2 (based upon image from CO2CRC). Reality: Developing the technologies and know-how to successfully capture and store CO 2 emissions will allow for a viable industry that will reduce the human contribution to atmospheric CO 2 levels. Myth: Carbon capture and storage is not a feasible way to reduce human CO 2 emissions. Department of Energy (DOE) is investigating five types of underground formations for geologic carbon storage:ĭOE’s Carbon Storage Program is conducting research and development (R&D) on CCS, developing Best Practice Manuals (BPMs) on topics. Geologic storage is defined as the placement of CO 2 into a subsurface formation so that it will remain safely and permanently stored. 
Suitable storage formations can occur in both onshore and offshore settings, and each type of geologic formation presents different opportunities and challenges. However, storage must be safe, environmentally sustainable, and cost-effective. WHAT IS CARBON CAPTURE AND STORAGE? Carbon storage diagram showing CO2 injection into a saline formation while producing brine for beneficial useĬarbon capture and storage (CCS) is the separation and capture of carbon dioxide (CO 2) from the emissions of industrial processes prior to release into the atmosphere and storage of the CO 2 in deep underground geologic formations.ĬCS enables industry to continue to operate while emitting fewer greenhouse gases (CHGs), making it a powerful tool for addressing mitigation of anthropogenic CO 2 in the atmosphere.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
